ORIGINAL
Sows’ parity and coconut oil postnatal supplement on piglets performance
Número de partos y la suplementación de aceite de coco para lechones recién nacidos
Luan S Santos,1 M.Sc, Fabiana R Caldara,2* Ph.D, Sivanilza T Machado,2 M.Sc, Irenilza A Nääs,2 Ph.D, Luciana Foppa,2 M.Sc, Rodrigo G Garcia,2 Ph.D, Rafael Moura,2 M.Sc, Simone P Machado,2 M.Sc.
1Universidade Estadual Paulista, Faculdade de Medicina Veterinária e Zootecnia, Botucatu, SP, Brazil.
2Universidade Federal da Grande Dourados, Faculdade de Ciências Agrárias, Dourados, MS, Brazil.
*Correspondencia: fabianacaldara@ufgd.edu.br
Received: August 2014; Accepted: December 2014.
Objective. The study aimed to evaluate the effect of sows’ of different parities and the supplement of coconut oil for piglets, on the development of litter. Materials and methods. A total of 51 sows of different parities and their 642 piglets were used in the trial. Each piglet was weighed and identified at birth in a sequential order. They were randomly distributed in two treatments (CG=control group and TG=test group). TG piglets had the first access to a dosage of 3.0 ml of coconut oil 12 hours after birth, and the second at 36 h after the first. Piglets were weighed at 21 days. In order to analyze the effect of the coconut oil supplement as a function of the weight at birth, piglets were grouped according to their weight (0.600 to 0.900 kg; 1.000 to 1.499kg; 1.500 to 1.999 kg; and 2.000 to 2.499 kg). Results. Sows parity affected the number and weight of born piglets. Sows in 4th, 5th and 7th parity had a larger litter than those from the 2nd parity. Sows from 2nd and 3rd parity had a lower number of piglets but heavier litter. No effect of the coconut oil supplement on neonatal piglets’ performance was found. Conclusions. The coconut meal was neither beneficial to neonatal piglets nor to those with low weight at birth, which usually present low body energy.
Key words: Energy sources, fatty acids, litter size, newborn animal (Source: CAB).
RESUMEN
Objetivo. El objetivo del estudio fue evaluar el efecto del número de partos y el suplemento de aceite de coco en lechones recien nacidos. Materiales y métodos. Se utilizaron 51 cerdas de diferentes partos y sus 642 lechones. Cada lechón se pesó y se identificó al nacer en orden secuencial. Fueron distribuidos aleatoriamente en dos grupos (CG = control y TG=tratamiento). Lechones TG tenían el primer acceso a una dosis de 3.0 ml de aceite de coco 12 horas después del nacimiento, y el segunda a las 36 h después de la primera. Los lechones fueron pesados a los 21 días. Con el fin de analizar el efecto de los suplementos de aceite de coco con relación al peso al nacer. Los lechones fueron agrupados de acuerdo con su peso (0.600 a 0.900 kg; 1.000 a 1.499 kg; 1.500 a 1.999 kg y 2.000 a 2.499 kg). Resultados. El número de partos afectó el peso y el número de lechones nacidos. Cerdas en cuarto, quinto y séptimo parto, tuvieron una camada mayor que las de segundo parto. Las cerdas de segundo y tercer parto tuvieron menor número de lechones y con peso mayor. No se encontró efecto del suplemento de aceite de coco en el desarrollo de los lechones recién nacidos. Conclusiones. Lo aceite de coco no es favorable para los lechones recién nacidos, tampoco para aquellos con el bajo peso al nacer, que normalmente presentan la energía corporal baja.
Palabras clave: ácidos grasos, animales recién nacidos, fuentes de energía, tamaño de la camada (Fuente: CAB).
Sows genetic development is intended for breeding, and to obtain prolific sows. Changes in management allowed the increase in a large number of weaned piglets per sow per year, from an average of 21-23 piglets (1) to near 28-30 weaned piglets per female per year (2). Consequently, there was an increase of problems related to birth weight, a large variability in weight between piglets (3), and competition for colostrum between pigs, increasing the challenge of the pig farmers in relation to management practices adopted after the farrowing. Besides this, sows of different age and different parities tend to have different litter size and weight at birth and weaning (4).
The piglet body energy stored at birth is present in the form of glycogen and fat. However, the available energy from glycogen and fat derivatives for newborn piglets is particularly low. Carbohydrates are the first source of energy used by newborn pig, and its absorption is significantly accelerated during the exposure to cold (5) leading to rapid depletion of glycogen. The amount of body fat is also particularly low at birth, and most of them are not available for oxidation. Thus, large litter with low weight neonatal piglets may result in high mortality rates in the first three days of life, mainly during cold weather (6). One way to overcome this issue may be to supply energy-rich supplements from sources of fast digestion and absorption to the piglets in their first days of life.
Triglycerides of medium chain fatty acids have 6 to 12 carbon atoms which are more easily digestible and metabolized than those of the long chain. The best response of lipids from coconut oil is related to its high number of medium chain fatty acids, which provides higher and faster absorption by the lymphatic system. This peculiarity distinguishes it from fatty acids of the other animal or vegetable fats. Thus, studies were carried out to assess the feasibility of using supplements such as coconut oil on blood homeostasis and improvement of the energy status of newborns piglets (7) obtaining conflicting results about to its benefits.
The present study aimed to evaluate the effects of parity and coconut oil supplement on the development of piglets after the farrowing.
MATERIALS AND METHODS
Animals and husbandry. The study was carried out in a commercial pig farm located at latitude 22°18’50’’ S and longitude 53°49’3’’ W, and an altitude of 360 m in Midwestern Brazil. The regional climate is Aw (according to Köpper classification), which means tropical climate with rain during the summer and dry season during winter. The farm had a total of 2300 sows genetically bred for prolific results. A total of 51 sows from different parity and their litter (624 piglets) were used in the trial. Sows were selected according to their integrity (locomotion, udder morphology, body score - 3 or 4 in a 5 scale). Primiparous sows were identified and analyzed separately. All females were kept under the same housing conditions, handling and feeding, and parturient sows had full assistance. After birth, the piglets were dried with paper towels and the umbilical cord cut and disinfected with iodine solution. Approximately 12 hours after the end of each birth piglets were individually weighed and identified with earrings numbered in sequential order. After identification, all of the piglets in the room the litters were equalized.
Experimental procedure. The piglets were distributed in a completely randomized design in two treatments (CG - control group and TG-test group). Each litter was present in the two treatments (50% of piglets in each group). Piglets from the test group received 3.0 ml of coconut oil 12 hours after birth and a second dose (3.0 ml) was provided 36 hours after the first. The coconut oil was administrated via oral. Piglets were weighed again individually at 21 days of age.
For evaluating the results of the coconut oil in the piglets weight gain, they were grouped according to their weight range 0.600 to 0.900 kg; 1.000 to 1.499 kg; 1.500 to 1.999 kg; and 2.000 to 2.4999 kg.
To determine the effect of the sows’ parity on the litter size and neonatal piglets’ weight sows parity sequence (1st to 7th) were considered the treatments. Analysis of variance was applied to data on weight of neonatal piglets and at 21 days of age, and F test was used for comparing the mean values. To evaluate the effects of parity the number of piglets and their neonatal weight data analysis of variance was applied, and Turkey’s test was used at a 95% level of confidence. To assess the correlations between parity, size and litter weight Pearson correlation analysis was applied. SAS (8) statistical program was used for computing data.
RESULTS
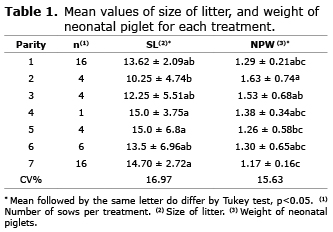
Effect of parity on the number and weight of neonatal piglets and at 21 days of age. In general, both variable’s size of litter (SL) and neonatal piglet weight (NPW) were influenced by parity. Smaller variations of both size of litter and weight of neonatal piglets were observed for 3rd and 4th parity. For the sows in 1st, 2nd, 5th, 6th and 7th parity a variation was found in the range of 15.36% to 26.86% for SL, and from 13.76 to 19.96% to NPW. Results suggest greater homogeneity in terms of both the size of litter and weight of neonatal piglets for sows between the 3rd and 4th gestation. There was an effect of parity in SL and NPW (Table 1).
Females of 4th, 5th, and 7th parity had larger litters than those from 2nd parity. Other sequence of parity did not differ. These results are in agreement with Bortolozzo and Wentz (9) who claim to have reduced the number of piglets born on the second parity in relation to the first, in several Brazilian farms.
Sows from 1st and 2nd parity presented longer time to return to estrus than sows from the 3rd parity (10). Sows from 1st and 2nd parity often eat less fodder during lactation (11) and, therefore, present stronger association between body weight loss during lactation and the weaning-to-estrus interval (12). Low ingestion of energy combined with high demand of energy for maintenance, milk production and body growth enhance the negative energy balance and catabolic stage in primiparous sows. This worsens the secretion of LH, the follicles maturation, and the occurrence of post weaning estrous. Sows which returned to estrus 7 to 10 days after weaning present decrease in farrowing rate and litter size, when compared to sow which return to estrous in 5 to 6 days (13). This may explain the reduction in size litter found in the present study, in sows, during 2nd parity.
Sows from 2nd and 3rd parity sequence had heavier piglets in litter, when compared to others, which is probably due to the fact that the litter had fewer piglets with higher individual weight. This could be confirmed by the correlations between the factors of order of birth, weight and litter size at birth. Higher parity increased the size of litter (0.2665; p<0.01), but decreased piglet’s weight at birth (-0.3144; p<0.01). And higher the birth order and the number of piglets per litter, the smaller the individual weight of neonatal piglets (-0.4334; p<0.01; Figure 1).
The piglets from the sows of 2nd, 3rd, and 4th sequence of parity were found within the ideal range of mean birth weight (14). However, those born from sows of 1st, 5th, and especially 7th parity presented neonatal weight considered low to critical. The age of sows interfere directly on the size of litter. Holanda et al (4) found quadratic effect of the age of sows on the size of litter, and the age in which this effect is maximized is 3.12 years. Higher size litters were found in sows aging 2.84 to 3.84 years, in the 5th and 6th parity, respectively. Quesnel et al (15) found an increase in the number of neonatal born alive piglets up to the 6th parity, and for the total size of litter up to the 7th parity. It should be expected an increase in the size of litter after the 2nd parity, and this occurs due to the increase in the ovulation rate and/or the survival of neonatal piglets. However, Vonnahme et al (16) found that parity sequence (2nd to 14th) did not affect ovulation rate and number of viable embryos and length of uterine horns sows at 25, 36 and 44 days of gestation, suggesting that the increase in embryos survival is responsible for the increase in litter size as the number of reproductive cycles increases.
The increase of prolificacy with increasing age of the sow has an indirect influence on individual neonatal piglet weight, assuming substantial value on the characteristics of variability of litter (4). The results found in the present study differ from those of Holanda et al (4) whose findings show a positive and linear effect of sows’ age at parity on the weight of neonatal piglets showing that the aging of sows led to heavier born piglets. In a study using sows in 1st to 10th parity sequence. Borges et al (17) found that parity affects the mean neonatal piglet weight, and the best findings were shown in sows, in the 3rd parity. However, the authors did not find a difference in relation to the number of born-alive piglets, dead neonatal piglets, or mummified embryos. Other researches also show an association between the size of litter and the weight of neonatal piglets, indicating a reduction of nearly 20 g in each additional piglet (4,18). This result of the size of litter on the weight of neonatal piglets confirms the results found by Kapell et al (19) and Bianchi (20). The authors show that large litter size with low neonatal weight leads to weak and less active piglets than others, implicating in high mortality before weaning.
Effect of coconut oil supplement in the weight gain of piglets. Mean neonatal piglets weight was 1.262 and 1.254 kg for the group test and control, respectively. Of all neonatal piglet weight, the higher frequency occurred between 0.9 and 1.5 kg (Figure 2). Similar values of mean average of weight of neonatal piglets were found by Fraga et al (21). Overall, there was no effect of supplementation of coconut oil on the weight of the piglets 21 days old, which were 5.080 kg and 5.020 kg for piglets supplemented with coconut oil and non-supplemented, respectively. The highest frequency of weight of piglets 21 days old occurred between the values of 4.2 to 6.0 kg suggesting that despite the oil supplementation did not contribute to the weight gain of the piglets, most of these had normal development during farrowing, as recommended by EMBRAPA (14). In a previous study, Hollanda et al (4) found slightly higher values for neonatal piglet weight (1.35 kg) and at 21 days old (5.6 kg).
When different weight categories were evaluated no beneficial effect of coconut oil supplementation on the performance of piglets (p>0.05) was detected, indicating that the supply of energy supplement showed no advantages on piglets weight gain, even for those of low neonatal weight, which typically have low body energy reserves (Table 2).
DISCUSSION
According to Empresa Brasileira De Pesquisa Agropecuária (EMBRAPA) (14), pig farmers must do their best to ensure that piglets obtain average daily weight gain of greater than 0.250 kg per day during farrowing. In the present study only the piglets belonging to the range of birth weight from 2.000 to 2.499 kg proven to increase this amount of weight. The body and colostrum carbohydrates are the main energy sources during the first hours, but the progressive decline of the respiratory rate during the first postnatal day provides evidence of the growing importance of lipids as an energy source during this time (22, 23). However, the low use of fatty acids as energy source during the neonatal period could be due to the limited potential for oxidation of fatty acids or other substrates of carbohydrates (24). The limited role of oxidation of fatty liver in newborn piglets acid level is low due to the mitochondrial activity and quantity of the enzyme 3-hydroxy- 3-methyl glutaryl - CoA synthase (25), which develops gradually with age. This fact could explain the findings in the present study. Thus, energy supplements to be provided in the first neonatal hours can cause some benefit when it is of digestible carbohydrates.
Parity affected the weight of piglets and the size of litter. Sows in second parity had smaller litter size, but with higher mean neonatal piglet weight. Energy diet of coconut oil in the first 48 hours did not increase piglets’ performance, even for those with low neonatal weight, which typically have very low body energy reserves.
REFERENCES
1. Tanghe S, Missotten J, Raes K, Vangeyte J, De Smet S. Diverse effects of linseed oil and fish oil in diets for sows on reproductive performance and pre-weaning growth of piglets. Livest Sci 2014, 164:109-118.
2. Antunes RC. Planejando a reposição de reprodutores (macho e fêmea) e impacto sobre a eficiência reprodutiva da granja. Rev Bras Reprod Anim 2007; 31: 41–46.
3. Fix JS, Cassady JP., Holl JW., Herring WO., Culbertson MS., See MT. Effect of piglet birth weight on survival and quality of commercial market swine. Livest Sci 2010, 132(1):98-106.
4. Holanda MCR, Barbosa SBP, Sampaio IBM, Santos ES, Santoro KR. Tamanho da leitegada e pesos médios, ao nascer e aos 21 dias de idade, de leitões da raça Large White. Arq Bras Med Vet Zoot 2005; 57: 539-544.
5. Hauptli L, Berto DA, Augusto RMN, Lo Tierzo V, Moraes KMCMT, Lucchesi L. Níveis de maltodextrina na dieta de leitões desmamados aos 21 dias. Acta Sci 2012; 34(3): 273-278.
6. Vasdal G, Østensen I, Melišová M, Bozděchová B, Illmann G, Andersen IL. Management routines at the time of farrowing–effects on teat success and postnatal piglet mortality from loose housed sows. Livest Sci 2011, 136(2):225-231.
7. Gebhart, K, Jensen D, Kitt S, Sonderman J, Hostetler C. Effect of split-suckling feeding coconut oil on piglet performance and survival. J Anim Sci 2011; 89:161.
8. SAS. Statistical Analysis System [CD-ROM]. Versión 9.0. Cary, NC, USA: SAS Inst, Inc; 2001.
9. Bortolozzo FP, Wentz I. Suinocultura em ação. A fêmea suína gestante. Porto Alegre: Gráfica da UFRGS, 2007.
10. Vargas AJ, Bernardi ML, Bortolozzo FP. Factors associated with return to estrus in first service swine females. Prev Vet Med 2009; 89: 75-80.
11. Kruse S, Traulsen I, Krieter J. Analysis of water, feed intake and performance of lactating sows. Livest Sci 2011; 135:177-183.
12. Thaker MYC, Bilkei G. Lactation weight loss influences subsequent reproductive performance of sows. Anim Reprod Sci 2005; 88:309-318
13. Poleze E, Bernardi ML, Amaral Filha WS, Wentz I, Bortolozzo FP. Consequences of variation in weaning-to-estrus interval on reproductive performance of swine females. Livest Sci 2006. 103:124-130.
14. Empresa Brasileira De Pesquisa Agropecuária(BR). Produção Suínos [Internet]. Concórdia (SC). EMBRAPA; 2000. [Accessed 2013 September 16]. URL Avaiable in: http://sistemasdeproducao.cnptia.embrapa.br/FontesHTML/Suinos/SPSuinos/manejoprodu.html
15. Quesnel H, Brossard L, Valancogne A, Quiniou N. Influence of some sow characteristics on within-litter variation of piglet birth weight. Animal 2008; 2(12):1842-1849.
16. Vonnahme KA, Wilson ME, Foxcroft GR, Ford SP. Impacts of conceptus survival in a commercial swine herd. J Anim Sci 2002; 80:553-559.
17. Borges VF, Bernardi ML, Bortolozzo FP, Wentz I. Perfil de natimortalidade de acordo com ordem de nascimento, peso e sexo de leitões. Arq Bras Med Vet Zootec 2008; 60(5):1234-1240
18. Pardo CE, Müller S, Bérard J, Kreuzer M, Bee G. Importance of average litter weight and individual birth weight for early postnatal performance and myofiber characteristics of progeny. Livest Sci 2013; 157(1):330-338.
19. Kapell DN, Ashworth CJ, Knap PW, Roehe R. Genetic parameters for piglet survival, litter size and birth weight or its variation within litter in sire and dam lines using Bayesian analysis. Livest Sci 2011; 135(2):215-224.
20. Bianchi I, Junior TL., Deschamps JC., Schneider A., Rabassa VR., Corrêa MN. Indicadores de desempenho relacionado ao parto de fêmeas suínas de primeiro e segundo partos. R Bras Zootec 2010; 39(6):1359-1362.
21. Fraga AB, Araújo Filho JT, Azevedo AP, Silva FL, Santana RS, Machado DFBP, Costa PPS. Peso médio do leitão, peso e tamanho de leitegada, natimortalidade e mortalidade em suínos no Estado de Alagoas. Rev Bras S Prod 2007; 8:354-363.
22. Yun J, Swan KM, Vienola K, Kim YY, Oliviero C, Peltoniemi OAT, Valros A. Farrowing environment has an impact on sow metabolic status and piglet colostrum intake in early lactation. Livest Sci 2014; 163:120-125.
23. Morise A, Louveau I, Le Huërou-Luron I. Growth and development of adipose tissue and gut and related endocrine status during early growth in the pig: impact of low birth weight. Animal 2008; 2:73-83.
24. Lallès JP, Bosi P, Smidt H, Stokes CR . Nutritional management of gut health in pigs around weaning. P Nutr Soc 2007; 66:260-268.
25. Duée PH, Pégorier JP, Quant PA, Herbin C, Kohl C, Girard J. Hepatic ketogenesis in newborn pigs is limited by low mitochondrial 3-hydroxy-3-methylglutaryl-CoA synthase activity. Biochem J 1994; 298:207–212.