COMUNICACIÓN BREVE
Serological evidence of Hepatitis E Virus infection in Antioquia, Colombia slaughtered pigs
Evidencia serológica de infección por el Virus de Hepatitis E en cerdos faenados en Antioquia, Colombia
Jorge Forero D,1* M.Sc, Cristian Gutiérrez V,1 M.Sc, Jaime Parra S,1 Ph.D, Guillermo Correa L,1 Ph.D, Berardo Rodríguez,2 Ph.D, Lina Gutiérrez B,3 Ph.D, Albeiro López-Herrera1 Ph.D.
1Universidad Nacional de Colombia, Facultad de Ciencias Agrarias. Departamento de Producción Animal, Grupo Biodiversidad y Genética Molecular BIOGEM. Calle 59A No.63-020, Autopista Norte, Bloque 50, Piso 3, Oficina 310. Medellín, Colombia.
2Universidad de Antioquia. Facultad de Ciencias Agrarias. Laboratorio de Patología Animal. Grupo de Investigación en Patobiología QUIRON. Carrera 75 No. 65-87. Bloque 47-134. Medellín. Colombia.
3Universidad Pontificia Bolivariana, Facultad de Medicina, Escuela de Ciencias de la Salud, Grupo Biología de Sistemas, Calle 78B N°72A-109, Bloque B. Medellín, Colombia.
*Correspondence: jforeroduarte@gmail.com
Received: Received: June 2014; Accepted: December 2014.
ABSTRACT
Objective. To detect the presence of specific antibodies against Hepatitis E virus (HEV) in pigs slaughtered in Antioquia, the department where the greatest amount of pork is produced and consumed in Colombia. Materials and methods. Between September 2011 and May 2012, blood samples from pigs were obtained in five slaughterhouses of Antioquia, four of them located in the Aburrá Valley subregion and other located in northern subregión. Serum were evaluated with a commercial ELISA kit for diagnosing HEV in humans but adapted to detect IgG and IgM antibodies in pigs. Results. A 100.0% seropositivity for IgG antibodies was found in 1000 samples evaluated, and 82.06% for IgM antibodies were found in 740 samples. Conclusions. These results indicate that pigs in slaughter age in Antioquia, and possibly in Colombia, have been exposed to HEV at some point in their production process and a high percentage of them can arrive to slaughterhouses with recent infection.
Key words: Antibodies, ELISA, Hepevirus, IgM, IgG, zoonosis (Source: DeCS).
RESUMEN
Objetivo. Detectar la presencia de anticuerpos específicos contra el virus de la Hepatitis E (HEV) en cerdos faenados en Antioquia, departamento donde se produce y consume la mayor cantidad de carne de cerdo en Colombia. Materiales y métodos. Entre septiembre de 2011 y mayo de 2012, se obtuvieron muestras de sangre de cerdos en cinco plantas de faenado, cuatro de ellas ubicadas en el Valle de Aburrá y una en la subregión Norte del departamento de Antioquia, las cuales fueron evaluadas mediante un kit de ELISA comercial para diagnóstico de HEV en humanos pero adaptado para la detección de anticuerpos tipo IgG e IgM en cerdos. Resultados. Se encontró una seropositividad de 100.0% para anticuerpos tipo IgG en 1000 muestras evaluadas y de 82.06% para anticuerpos tipo IgM en 740 muestras. Una muestra de heces positiva para la detección del genoma HEV es similar al genotipo 3 encontrada en Estados Unidos. Conclusiones. Estos resultados indican que los cerdos en edad de faenado en Antioquia y posiblemente en Colombia, han tenido exposición a HEV del, un virus zoonótico emergente a nivel mundial, en algún momento de su proceso productivo.
Palabras clave: Anticuerpos, ELISA, Hepevirus, IgM, IgG, zoonosis (Fuente: DeCS).
INTRODUCTION
Hepatitis E is an acute liver disease in humans caused by the hepatitis E virus (HEV). Although this type of hepatitis has a global distribution, in some countries of Africa and Asia outbreaks associated with contamination of water sources for consumption are presented, while in other regions such as Europe and the United States there are sporadic outbreaks associated with the consumption of raw or undercooked food (1, 2). It is estimated that every year around the world, 20 million humans are infected, with about three million cases of acute hepatitis and 57,000 deaths related to Hepatitis E (1.3). Most of the time the disease is self-limiting in humans; however, it can be extremely serious for pregnant women, and the mortality rate for this population may exceed 20% (4).
There are four major HEV genotypes that are recognized in mammals. Genotypes 1 and 2 are restricted to humans while genotypes 3 and 4 can infect, in addition to humans, other species, mainly domestic pigs (3). Genotype 1 is found mainly in India, while genotype 2 has been detected in several countries of Africa and in Mexico. Genotype 3 is found around the world, while genotype 4 is often found in Asian countries like China and Japan (1-3).
In Latin America, the first serologic evidence of the virus in humans was found in Venezuela in 1994; later, in other research on the seroprevalence of HEV in countries in the region, varying percentages were found in the different human populations studied, none of which exceed 20% (5). Additionally, the viral genome has been detected in humans in Argentina, Brazil, Bolivia and Uruguay, where genotype 3 has been identified; however, in Venezuela and Argentina genotype 1 has been found (5).
In addition to the findings in humans, serologic evidence of infection has been found in other species such as cattle, dogs and chickens (6), and the viral genome in wild boars, deer, rabbits, rats and bats (2,7,8) but it is still unclear what role these species play in the epidemiology of the infection. On the other hand, the increased risk of infection in swine handlers (9), the high genetic identity between swine and human HEV viral sequences (10,11), and the experimental infection with swine genotypes to nonhuman primates, are findings which identified the pig as the viral reservoir of infection (2,10,11).
Serological studies in swine show a wide distribution of the virus in herds around the world. In the United States seropositivity reached 41% (12), 90% in New Zealand (13), 100% in Mexico (14) and 98% in Spain (15). Studies in Latin America have reported the circulation of HEV in pigs in Argentina (16), Brazil (17), Chile (18), Costa Rica (19) and Mexico (14).
Pig production is an important part of the livestock sector in Colombia, with a positive impact on the economy, not only by promoting the livestock sector, but as a source of employment in the country (20). Antioquia is the department in Colombia with the greatest production and consumption of pork, as it provides about 45% of total production in the country (20).
Hepatitis E is considered a global public health problem; however, in Colombia, despite some isolated efforts, the status for HEV in the population is unknown, as well as the presence of specific antibodies in pigs bred in the country. The aim of this study was to determine the presence of IgG and IgM antibodies in pigs slaughtered in the main slaughterhouses in the department of Antioquia.
MATERIALS AND METHODS
Ethical aspects. This study was conducted in accordance with protocols approved by the Ethics Committee of the Universidad Nacional of Colombia, Medellin (Act CEMED-226, of October 14, 2011).
Sampling and size of sample. Sampling was conducted with slaughtered pigs between September 2011 and May 2012 at five slaughterhouses, four of them located in the Aburrá Valley and in the northern subregion of the department of Antioquia, which were identified in the present study with letters A to E (randomized) in order to maintain confidentiality. The selected plants process the majority of the swine production in Antioquia. The average number of animals slaughtered per day during the six months prior to the start of the study was 450, 200, 400, 800 and 400 animals in plants A, B, C, D and E, respectively.
Based on the sample size required to estimate seropositivity in Antioquia with a confidence level of 95% and a maximum error of 3%, a total of 1,000 samples were evaluated, allocating per plant the average daily amount of animals processed over the past six months. Within each slaughterhouse a sampling by quotas was performed, where each quota was calculated in proportion to the number of animals slaughtered in the subregion of origin. In each slaughterhouse, the pigs were selected systematically, with a random start.
Collecting information. The sampled animals were identified by place of origin, classified as healthy, and approved for slaughter and commercialization, according to the pre and post-mortem health inspection done by the responsible veterinarians at each processing plant.
Serum samples. Blood samples were obtained on the slaughtering line after desensitizing the animal, at the time of bleeding. Approximately 10ml of blood was taken per animal in sterile 15 ml tubes. The samples were put in cold storage to be transported to the laboratory of Animal Biotechnology of the Universidad Nacional of Colombia, in Medellin, where they were immediately centrifuged at 1500 rpm for 10 minutes to obtain the serum, which was stored at -20°C until analyzed.
Indirect ELISA immunoenzymatic analysis to detect specific IgG and IgM antibodies against HEV. To detect IgG antibodies against HEV in pigs, the commercial kit of indirect ELISA HEV-IGAB (Dia.Pro-Diagnostic Bioprobes, Italy) for dual detection of IgG and IgM antibodies in human serum was used, replacing the anti-human conjugate with a conjugated goat antibody anti-IgG for swine (Thermo Fisher Scientific Inc, USA) according to the methodology used by other researchers (17,21-23) at the dilution recommended by the manufacturer (1:5000). As a negative swine control (NPC), a serum of newborn piglets (in triplicate) that did not ingest colostrum was used, to avoid the presence of maternal antibodies; and as a positive control (CPP) a sample of the study that showed high OD values in the ELISA was employed. In each of the dishes that swine serum was evaluated, and for purposes of internal quality control for each test, the controls recommended and provided by the manufacturer were included: three negative controls (NC), a positive control (CP) and two calibrators (C) evaluated with the anti-human conjugate in the case.
To detect IgM antibodies to HEV in pigs, the same strategy previously mentioned was used, but a commercial indirect ELISA kit HEV-IgM (Dia.Pro-Diagnostic Bioprobes, Italy) was used, replacing the human anti-IgM conjugate with a goat antibody conjugate anti-IgM for swine (ScientificInc Thermo Fisher, USA) in a 1:5000 dilution recommended by the manufacturer. A serum sample from a newborn piglet was used as CNP with the above mentioned characteristics, and as CPP a serum sample of a pig with molecular detection of viral shedding in feces affected by PCR for the ORF1 region of HEV. For purposes of internal quality control of each test, controls that are recommended and provided by the manufacturer were included: three CN and one CP evaluated with the anti-human conjugate of the case.
The microplate of the cases used in this study were coated with specific synthetic HEV antigens derived from the ORF2 and ORF3 regions of the 4 genotypes as described by the manufacturer. OD readings were obtained on an ELISA reader (Biotek, USA) with a 450 nm filter.
Viability of the indirect modified ELISA test to detect IgG and IgM antibodies. To ensure the viability of the modified conjugated immunoassays with the anti-swine conjugates, initially a positive swine serum sample for IgG anti-HEV characterized and evaluated in a previous study, kindly donated by Dr. Munné (National Reference Laboratory for viral hepatitis INEI ANLIS “Carlos Malbrán”, Argentina), (17). This serum was positive with the selected cutoff point in this project, but preliminary tests indicated that the optical density reading at 450 nm (OD 450) for the Argentine positive serum were two times lower than DO450 of some Colombian samples (data not shown); for this reason, and because the volume of the donated positive serum was insufficient to use as a positive control in all assays, the CPP and CNP described above were used.
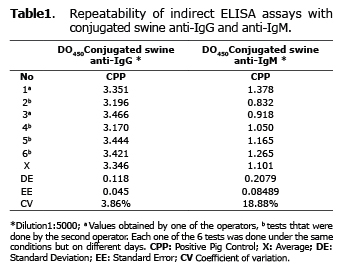
To assess the repeatability of ELISA assays modified anti- IgG and IgM in swine, six successive tests on different days, with two different operators and under the same laboratory conditions, were performed and a coefficient of variation (CV) was obtained with less than 20% gross absorbance values, so it was considered that the trials had adequate levels of repeatability (23,24).
Estimating the value of the cut for positive IgG and IgM antibodies. Obtaining the cutoff for positivity of the IgG and IgM antibodies was performed based on Chebyshev inequality (25), which states that, regardless of the probability distribution of a random variable, no more than (1/k2) % of those distribution values found over k standard deviations from the mean. To control the probability of false positives, the maximum tolerable probability was set at 0.0102, which corresponds to the cutoff given by the average value of the readings of the negative controls plus seven standard deviations. Using this criterion, the cutoffs were 0.220 and 0.545 for IgG and IgM, respectively.
Statistical methods. Repeatability analysis was based on the evaluation of the DO results obtained by the CV, standard deviation (SD) and standard error (SE). Control charts of Levey Jennings were constructed from DO data using the Excel program. To analyze HEV seropositivity in pigs in the slaughterhouses and by geographic subregion, the frequencies of data were estimated, the confidence intervals (with a confidence level of 95%) and differences between proportions (based on Fisher’s exact test), with a confidence level of 95%; they are considered significant at p <0.05. All analysis was performed using GraphPad Prism tools 5 (GraphPad Software, Inc. La Jolla, USA).
RESULTS
Repeatability and consistency of modified indirect ELISA tests to detect IgG and IgM antibodies. After determining the optimal dilution of the conjugate and the test samples, the repeatability of the test was evaluated by evaluating the CPP in six independent trials with two different operators. The CV values obtained for the CPP were 3.86% and 18.88% for IgG and IgM respectively (Table 1).
The DO450 values of the CPP in each of the tests for the two antibodies were controlled throughout the study by means of Levey-Jennings graphics as shown in figure 1, the values obtained in each one of the determinations did not exceed the average value in more than two DE both for IgG as well as IgM. Similarly, it is noted that all the kit controls operated within the parameters defined by the manufacturer in each of the tests (data not shown).
Frequency of specific HEV antibodies in swine serum from Antioquia. The frequency of positivity found in the serum of pigs sampled in the slaughterhouses in Antioquia, with cut off points estimated in the study to detect IgG and IgM, was 100% and 82.06%, respectively.
IgM positivity in each slaughterhouse were similar and ranged between 74.49 and 87.98% (Table 2), with a significant difference between Plant A and Plant B and E with values at p=0.0067 and p=0.0042, respectively. By analyzing the origin of the swine, it was seen that the sampled animals came from six of the nine subregions of the department of Antioquia (Figure 2). The percentages of positive pigs and confidence intervals for detecting IgM anti HEV antibodies by subregion of origin are shown in table 2. The percentage of pigs positive for IgM ranged from 77.78% for the West subregion and 88.89% for the Northeast subregion (Figure 2), but no significant difference was found between the regions analyzed.
DISCUSSION
Many of the reported tests for the detection of anti HEV antibodies in pigs and other animals use modified commercial immunoassays to detect antibodies of the animal of interest (8,16); however, there are few reports about the reliability of such modifications.
Evaluating the repeatability of a diagnostic test is one of the most important parameters to determine its reliability (23). The CV for the CPP for IgG and IgM obtained from the modifications to the commercial test, on different days and with different operators, were within the acceptable ranges for a diagnostic test (25); however, it is necessary in future studies to reaffirm the data obtained from the modified assay by evaluating the serum with a specific validated case for detecting antibodies in pigs and establishing the diagnostic sensitivity and specificity of the test.
For any serological test it is essential to determine the cutoff, which is defined as the level of activity of antibody that discerns the positive status of the negative of an animal for a particular biological analyte (26). The most appropriate way to calculate it is by using the reference serum of known status (25,26). In the absence of reference serum, often rough estimates are used by evaluating the distribution frequencies of the OD of the antibody to be detected in a large population or using the average OD values of the negative controls are used and adding various DS (27).
In this study a distribution of bimodal frequencies was obtained in the case of detecting IgG antibodies, for which a first approach to the cutting point was made using a statistical tool to resolve finite mixtures (8,24,26) (data not shown).
From that it was possible to estimate a cut-off point of 3.31, which yielded a seropositivity of 26% (data not shown). This value may overestimate the specificity of the test, since DO450 values, for example of 1.6, would be declared negative, increasing the risk of having a proportion of false negatives. Therefore, the cutoff points were determined using a second approach that consisted of establishing the cut-off value based on the average values of DO450 of the negative controls plus 7 DS (27), decreasing the probability of false negatives, methodology which allows a more realistic approximation of the situation of virus positivity for pigs in this study.
In this context, the results shown indicated that all animals that arrived at slaughterhouses in Antioquia in the study period (the average slaughter age in Antioquia is 22 weeks (28)), were exposed to HEV at some point in the production process. Studies of natural infection in pigs show that these are infected at some point after the titers of maternal antibodies for IgG have decreased (between 8-10 weeks of life) approximately between week 14 to 17. Their IgG antibodies can be detected in a high proportion of pigs at slaughter age (29), which is consistent with that observed in this study.
Unlike IgG antibodies, IgM antibodies in naturally infected pigs decreased 4 to 7 weeks after infection, that is, between 15-17 weeks of age in a large amount of positive animals, but at slaughter age this ratio tends to decrease (29). The reason why in this study the proportion of pigs that are seropositive for IgM at slaughter age was high is unknown, but it could be related to the handling of animals in herds (density of animals on the premises, biosanitary management on farms, type of production, level of technology etc.) or greater pathogenicity of the virus circulating in the region.
In addition to this, the positivity for IgM antibodies suggests recent infection and although viremia and the excretion period are relatively short in pigs (17), it is possible that a significant proportion of animals that were the environment were excreting the virus at the time of slaughter.
The observed differences in positivity for IgM in slaughterhouses could be related to the flow of animals from the subregions with greater positivity, since it is unlikely to have increased in IgM production in the short stay at the slaughterhouse. HEV infection in pigs produced no apparent clinical signs or caused changes that were visible in the health inspections carried out in the slaughterhouse (29); however, given the high rates of positivity found, it is possible that the pigs may be have associated microscopic liver lesions.
Pork meat and in particular livers are major part of the diet in some sectors of the Colombian population. The high percentage of recent infection in pigs at slaughterhouses suggests that livers sold in Antioquia may be contaminated with the virus, so it is advisable to take care to consume them after an adequate cooking process, since it has been shown that the virus is inactivated at temperatures above 70°C held for 10 minutes (30).
The high seropositivity of IgG and IgM antibodies found in this study are greatly important to public animal health since it indicates that the virus is circulating on swine herds of department of Antioquia (and probably the country), and that a significant proportion of animals at slaughter age could be shedding the virus into the environment, which should draw the attention of the scientific community to study the dynamics of the virus in the country, and the health authorities to implement assessment and control measures to prevent the transmission of HEV to the swine production in the country.
In conclusion, as far as the research group knows, this is the first serologic evidence of the presence of HEV in pigs at slaughtering age in Colombia, and although the values of antibody seropositivity in the animals in the study was high, it is necessary to investigate individual conditions that contribute to pigs for human consumption in Antioquia being exposed to the virus and therefore generate control measures to prevent the spread of the virus in swine production in the department.
REFERENCES
1. Aggarwal R, Naik S. Epidemiology of hepatitis E: current status. J Gastroenterol Hepatol 2009;24(9): 1484-93.
2. Meng XJ. Zoonotic and foodborne transmission of hepatitis E virus. Semin Liver Dis 2013;33(1):41-49.
3. Kumar S, Subhadra S, Singh B, Panda BK. Hepatitis E virus: the current scenario. Int J Infect Dis 2013; 17(4):e228-e233.
4. Kumar A, Beniwal M, Kar P, Sharma JB, Murthy NS. Hepatitis E in pregnancy. Int J Gynaecol Obstet 2004;85(3):240-244.
5. Echevarría JM, González JE, Lewis-XimenezLL, Dos Santos DR, Munné MS, Pinto MA,et al.Hepatitis E virus infection in Latin America: a review. J Med Virol 2013; 85(6):1037-1045.
6. Vitral CL, Alves M, Lewis-Ximenez LL, Khudyakov YE, Lopes dos Santos DR, Gaspar AM. Serological evidence of hepatitis E virus infection in different animal species from the Southeast of Brazil. Mem Inst Oswaldo Cruz 2005;100(2):117–122.
7. Cossaboom CM, Córdoba L, Cao D, Ni YY, Meng XJ. Complete genome sequence of hepatitis E virus from rabbits in the United States. J Virol 2012; 86(23):13124-13125
8. Rutjes SA, Lodder-Verschoor F, Lodder WJ, van der Giessen J, Reesink H, Bouwknegt M, et al. Seroprevalence and molecular detection of hepatitis E virus in wild boar and red deer in The Netherlands. J Virol Methods 2010; 168(1-2):197-206.
9. Pourpongporn P, Samransurp K, Rojanasang P, Wiwattanakul S, Srisurapanon S. The prevalence of anti-hepatitis E in occupational risk groups. J Med Assoc Thai 2009; 92(Suppl 3):S38–S42
10. Ma H, Geng Y, Li Z, Harrison TJ, Huang W, Wang Y. Analysis of the complete genome sequences of one swine and two human hepatitis E virus genotype 4 strains isolated in Beijing, China. Infect Genet Evol 2013; 18:42-47.
11. Wang H, He Y, Shen Q, Wang X, Yang S, Cui L, et al. Complete genome sequence of the genotype 4 hepatitis E virus strain prevalent in swine in Jiangsu Province, China, reveals a close relationship with that from the human population in this area. J Virol 2012; 86(15):8334-8335.
12. Dong C, Meng J, Dai X, Liang JH, Feagins AR, Meng XJ, et al. Restricted enzooticity of hepatitis E virus genotypes 1 to 4 in the United States. J Clin Microbiol 2011; 49(12):4164-4172.
13. Garkavenko O, Obriadina A, Meng J, Anderson DA, Benard HJ, Schroeder BA, et al. Detection and characterisation of swine hepatitis E virus in New Zealand. J Med Virol 2001; 65(3):525–529.
14. Cooper K, Huang FF, Batista L, Rayo CD, Bezanilla JC, Toth TE, et al. Identification of genotype 3 hepatitis E virus (HEV) in serum and fecal samples from pigs in Thailand and Mexico. J Clin Microbiol 2005; 43(4):1684-1688.
15. Seminati C, Mateu E, Peralta B, de Deus N, Martin M. Distribution of hepatitis E virus infection and its prevalence in pigs on commercial farms in Spain. Vet J 2008 175(1):130–132.
16. Munné MS, Vladimirsky S, Otegui L, Castro R, Brajterman L, Soto S, et al. Identification of the first strain of swine hepatitis E virus in South America and prevalence of anti-HEV antibodies in swine in Argentina. J Med Virol 2006; 78(12):1579-1583.
17. Paiva HH,Tzaneva V, Haddad R,Yokosawa J. Molecular characterization of swine hepatitis E virus from southeastern Brazil. Braz J Microbiol. 2007; 38(4):693-698.
18. Ibarra VH, Riedemann GS, Reinhardt VG, Calvo AM. Presence of anti hepatitis E virus antibodies in swine: is it an animal reservoir for hepatitis. Rev Med Chil 2007; 135(8):997-1001.
19. Kase JA, Correa MT, Luna C,Sobsey MD.Isolation, detection and characterization of swine hepatitis E virus from herds in Costa Rica. Int J Environ Health Res 2008;18(3):165-176.
20. Agronet.com [sede web] Bogotá: Departamento Administrativo Nacional de Estadística (DANE); 23 de octubre de 2012[Acceso 15 de diciembre de 2013] Boletín mensual INSUMOS Y FACTORES DE PRODUCCIóN: La carne de cerdo en el mundo. URL Disponible en: http://www.agronet.gov.co
21. Huang FF, Haqshenas G, Guenette DK,Halbur PG, Schommer SK, Pierson FW, et al. Detection by reverse transcription-PCR and genetic characterization of field isolates of swine hepatitis E virus from pigs in different geographic regions of the United States. J Clin Microbiol 2002; 40(4):1326–1332.
22. Leblanc D, Ward P, Gagné MJ, Poitras E, Müller P, Trottier YL, et al. Presence of hepatitis E virus in a naturally infected swine herd from nursery to slaughter. Int J Food Microbiol 2007; 117(2):160-166.
23. Jacobson RH. Validation of serological assays for diagnosis of infectious diseases. Rev Sci Tech 1998; 17:469-526.
24. Chernick, MR. The Essentials of Biostatistics for Physicians, Nurses, and Clinicians. New Jersey. John Wiley & Sons. 2011. pp.49–50.
25. OIE [sede web]. Manual of Diagnostic Tests and Vaccines for Terrestrial Animals 2013.Principles and methods of validation of diagnostic assays for infectious diseases. 2013 [Acceso 15 diciembre 2013] URL Disponible en: http://www.oie.int/fileadmin/Home/fr/Health_standards/tahm/1.01.05_VALIDATION.pdf
26. Crowther JR. The ELISA guidebook. 2nd Edition. New York; Human press: 2008.
27 Mizuo H, Suzuki K, Takikawa Y, Sugai Y, Tokita H, Akahane Y, et al.Polyphyletic strains of hepatitis E virus are responsible for sporadic cases of acute hepatitis in Japan. J Clin Microbiol 2002;40(9):3209-3218.
28. Díaz CA, Rodríguez MN, Vera VJ, Ramirez G, Casas GA, Mogollón JD. Caracterización de los sistemas de producción porcina en las principales regiones porcícolas colombianas. Rev Colomb Cienc Pecu 2011; 24(2): 131-144.
29. de Deus N, Casas M, Peralta B,Nofrarías M, Pina S, Martín M,et al. Hepatitis E virus infection dynamics and organic distribution in naturally infected pigs in a farrow-to-finish farm. Vet Microbiol 2008; 13(1-2):19-28.
30. Yunoki M, Yamamoto S, Tanaka H, Nishigaki H, Tanaka Y, Nishida A, et al. Extent of hepatitis E virus elimination is affected by stabilizers present in plasma products and pore size of nanofilters. Vox Sang 2008; 95(2):94-100.