Una revisión sumaria sobre algunos virus veterinarios importantes en las Américas
A concise review on certain important veterinary viruses in the Americas
Mostrar biografía de los autores
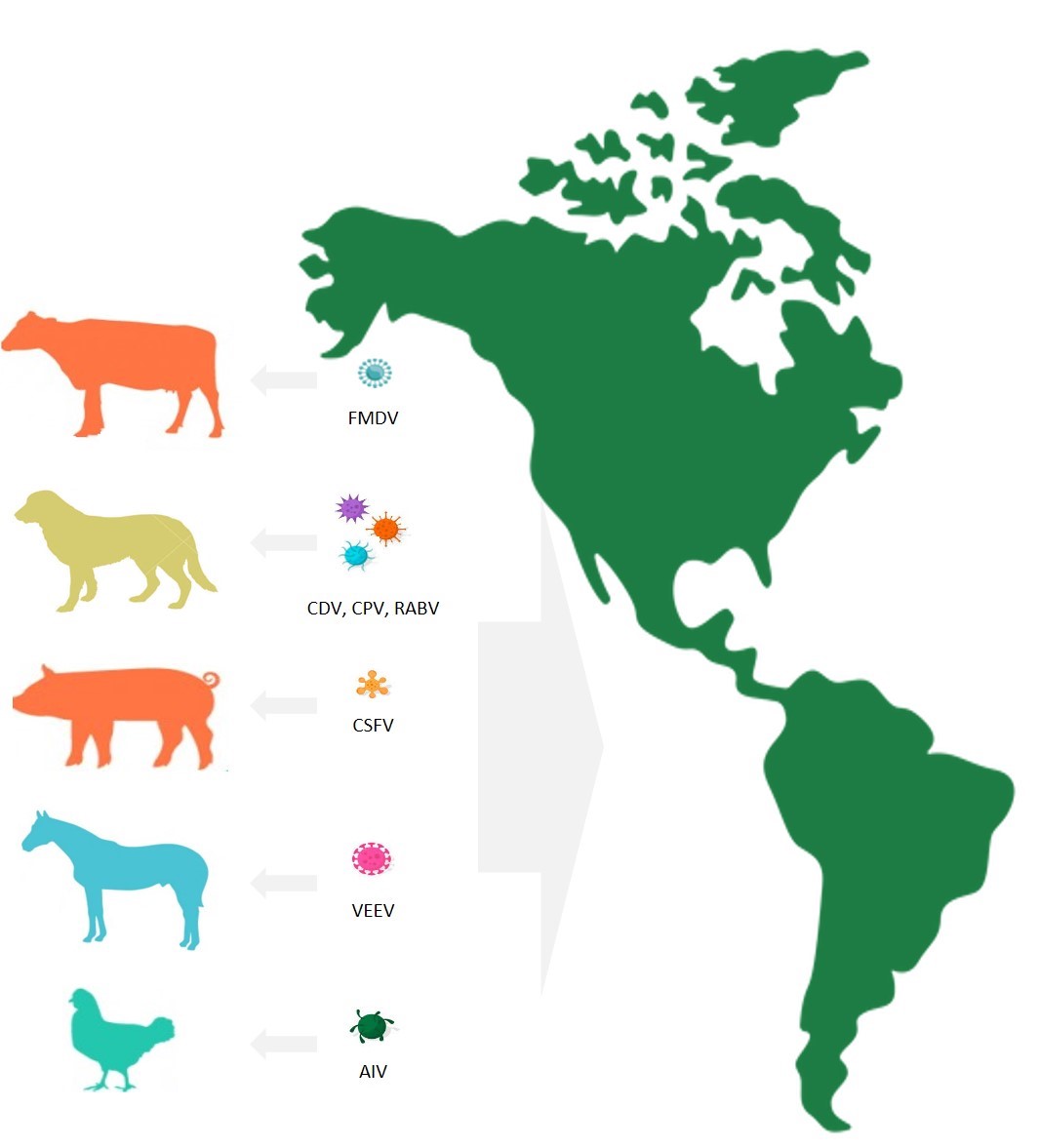
Los animales se han convertido en miembros esenciales de nuestra Sociedad, con roles que incluyen compañía, bienestar y terapia para algunas enfermedades, así como fuente de alimento para muchas poblaciones alrededor del mundo. Los animales destinados para el consumo humano como cerdos y bovinos, así como los animales de compañía, especialmente los gatos y perros están constantemente amenazados por múltiples agentes virales, lo cual pone en riesgo a los propietarios de mascotas y amenaza la seguridad alimentaria en las regiones. Sabiendo que el control o la erradicación es un problema complejo que involucra diversos aspectos, existe un éxito limitado respecto a esto para enfermedades virales tales como la fiebre aftosa, rabia, la peste porcina clásica o africana. Esta revisión tiene como objetivo mostrar las características más importantes, en el contexto epidemiológico, de virus de animales de producción, virus reemergentes que afectan animales de compañía y virus zoonóticos emergentes y reemergentes. Debido a que los patógenos virales afectan poblaciones animales y el bienestar humano, existe la necesidad de incrementar los esfuerzos en el control, monitoreo y erradicación de estos tanto de los animales de producción como en los animales de compañía. Por lo cual, las siguientes secciones contribuyen a mejorar el entendimiento de estos agentes virales y diseñar acciones de control en las Américas.
Visitas del artículo 1524 | Visitas PDF
Descargas
- Walsh F. Human-animal bonds I: the relational significance of companion animals. Fam Process. 2009; 48(4):462-480. https://doi.org/10.1111/j.1545-5300.2009.01296.x
- Christou L. The global burden of bacterial and viral zoonotic infections. Clin Microbiol Infect. 2011; 17(3):326-330. https://doi.org/10.1111/j.1469-0691.2010.03441.x
- Reperant L, Brown I, Haenen O, de Jong M, Osterhaus AD, Papa A, et al. Companion Animals as a Source of Viruses for Human Beings and Food Production Animals. Journal of Comparative Pathology. 2016; 155(1):S41-S53. https://doi.org/10.1016/j.jcpa.2016.07.006
- Ruiz-Saenz J, Goez Y, Tabares W, Lopez-Herrera A. Cellular receptors for foot and mouth disease virus. Intervirology. 2009; 52(4):201-212. https://doi.org/10.1159/000226121
- Robinson L, Knight-Jones TJ, Charleston B, Rodriguez LL, Gay CG, Sumption KJ, et al. Global Foot-and-Mouth Disease Research Update and Gap Analysis: 7 - Pathogenesis and Molecular Biology. Transbound Emerg Dis. 2016; 63(Suppl 1):63-71. https://doi.org/10.1111/tbed.12520
- Clavijo A, Sanchez-Vazquez MJ, Buzanovsky LP, Martini M, Pompei JC, Cosivi O. Current Status and Future Prospects to Achieve Foot-and-Mouth Disease Eradication in South America. Transbound Emerg Dis. 2017; 64(1):31-36. https://doi.org/10.1111/tbed.12345
- Knight-Jones TJ, Robinson L, Charleston B, Rodriguez LL, Gay CG, Sumption KJ, et al. Global Foot-and-Mouth Disease Research Update and Gap Analysis: 1 - Overview of Global Status and Research Needs. Transbound Emerg Dis. 2016; 63(Suppl 1):3-13. https://doi.org/10.1111/tbed.12528
- Knight-Jones TJ, Robinson L, Charleston B, Rodriguez LL, Gay CG, Sumption KJ, et al. Global Foot-and-Mouth Disease Research Update and Gap Analysis: 4 - Diagnostics. Transbound Emerg Dis. 2016; 63(Suppl 1):42-48. https://doi.org/10.1111/tbed.12523
- Knight-Jones TJ, Robinson L, Charleston B, Rodriguez LL, Gay CG, Sumption KJ, et al. Global Foot-and-Mouth Disease Research Update and Gap Analysis: 2 - Epidemiology, Wildlife and Economics. Transbound Emerg Dis. 2016; 63(Suppl 1):14-29. https://doi.org/10.1111/tbed.12522
- PAHO. Informe de situación de los programas de erradicación de la fiebre aftosa en Sudamérica y Panamá, año 2018. Rio de Janeiro, Brazil: Pan American Health Organization; 2019.
- Prempeh H, Smith R, Muller B. Foot and mouth disease: the human consequences. The health consequences are slight, the economic ones huge. BMJ. 2001; 322(7286):565-566. https://doi.org/10.1136/bmj.322.7286.565
- Maclachlan NJ, Dubovi EJ. Fenner’s veterinary virology: Academic press; London, UK; 2010.
- Chan JF, To KK, Chen H, Yuen KY. Cross-species transmission and emergence of novel viruses from birds. Current Opinion in Virology. 2015; 10:63-69. https://doi.org/10.1016/j.coviro.2015.01.006
- Parrish CR, Murcia PR, Holmes EC. Influenza virus reservoirs and intermediate hosts: dogs, horses, and new possibilities for influenza virus exposure of humans. Journal of Virology. 2015; 89(6):2990-2994. https://doi.org/10.1128/JVI.03146-14
- Tong S, Li Y, Rivailler P, Conrardy C, Castillo DA, Chen LM, et al. A distinct lineage of influenza A virus from bats. Proc Natl Acad Sci U S A. 2012; 109(11):4269-4274. https://doi.org/10.1073/pnas.1116200109
- Tong S, Zhu X, Li Y, Shi M, Zhang J, Bourgeois M, et al. New world bats harbor diverse influenza A viruses. PLoS Pathog. 2013; 9(10):e1003657. https://doi.org/10.1371/journal.ppat.1003657
- Wille M, Robertson GJ, Whitney H, Ojkic D, Lang AS. Reassortment of American and Eurasian genes in an influenza A virus isolated from a great black-backed gull (Larus marinus), a species demonstrated to move between these regions. Arch Virol. 2011; 156(1):107-115. https://doi.org/10.1007/s00705-010-0839-1
- Alexander DJ. An overview of the epidemiology of avian influenza. Vaccine. 2007; 25(30):5637-5644. https://doi.org/10.1016/j.vaccine.2006.10.051
- Abdelwhab EM, Hafez HM. An overview of the epidemic of highly pathogenic H5N1 avian influenza virus in Egypt: epidemiology and control challenges. Epidemiol Infect. 2011; 139(5):647-657. https://doi.org/10.1017/S0950268810003122
- Setiawaty V, Dharmayanti NL, Misriyah, Pawestri HA, Azhar M, Tallis G, et al. Avian Influenza A(H5N1) Virus Outbreak Investigation: Application of the FAO-OIE-WHO Four-way Linking Framework in Indonesia. Zoonoses and public health; 2014. https://doi.org/10.1111/zph.12158
- WHO. Cumulative number of confirmed human cases for avian influenza A(H5N1) reported to WHO, 2003-2019. París: World Health Organization: WHO; 2019. https://www.who.int/influenza/human_animal_interface/H5N1_cumulative_table_archives/en/
- Kilbourne ED. Influenza pandemics of the 20th century. Emerg Infect Dis. 2006; 12(1):9-14. https://doi.org/10.3201/eid1201.051254
- de Jong MD, Hien TT. Avian influenza A (H5N1). J Clin Virol. 2006; 35(1):2-13. https://doi.org/10.1016/j.jcv.2005.09.002
- Dawood FS, Iuliano AD, Reed C, Meltzer MI, Shay DK, Cheng PY, et al. Estimated global mortality associated with the first 12 months of 2009 pandemic influenza A H1N1 virus circulation: a modelling study. Lancet Infect Dis. 2012; 12(9):687-695. https://doi.org/10.1016/S1473-3099(12)70121-4
- Williams GW, Anderson DP. The Latin American Livestock Industry: Growth and Challenges. Choices. 2020; 34(316-2020-617):1-11. https://www.choicesmagazine.org/choices-magazine/submitted-articles/the-latin-american-livestock-industry-growth-and-challenges
- Marquez MA. Control and prevention of Avian Influenza, a Latin American perspective. AviNews International Magazine. 2020. https://avicultura.info/en/control-and-prevention-of-avian-influenza-a-latin-american-perspective/
- Afanador-Villamizar A, Gomez-Romero C, Diaz A, Ruiz-Saenz J. Avian influenza in Latin America: A systematic review of serological and molecular studies from 2000-2015. PLoS One. 2017; 12(6):e0179573. https://doi.org/10.1371/journal.pone.0179573
- Senne DA. Avian influenza in North and South America, 2002-2005. Avian diseases. 2007; 51(S1):167-173. https://doi.org/10.1637/7621-042606R1.1
- Suarez DL, Senne DA, Banks J, Brown IH, Essen SC, Lee C-W, et al. Recombination resulting in virulence shift in avian influenza outbreak, Chile. Emerg Infect Dis. 2004; 10(4):693-699. https://dx.doi.org/10.3201%2Feid1004.030396
- OIE. OIE Situation Report for Highly Pathogenic Avian Influenza: World Organisation for Animal Health; 2020. https://www.oie.int/en/animal-health-in-the-world/update-on-avian-influenza/2020/
- Mishra A, Vijayakumar P, Raut AA. Emerging avian influenza infections: Current understanding of innate immune response and molecular pathogenesis. Int Rev Immunol. 2017; 36(2):89-107. https://doi.org/10.1080/08830185.2017.1291640
- Wainwright S, Trevennec C, Claes F, Vargas-Terán M, Martin V, Lubrotha J. Highly pathogenic avian influenza in Mexico (H7N3). Food and Agriculture Organization; 2012. http://www.fao.org/docrep/016/an395e/an395e.pdf
- Beldomenico PM, Uhart M. Ecoepidemiología de los virus de influenza aviar. FAVE Sección Ciencias Veterinarias. 2008; 7(1/2):23-40. https://doi.org/10.14409/favecv.v7i1/2
- Pepin KM, Spackman E, Brown JD, Pabilonia KL, Garber LP, Weaver JT, Riley S. Using quantitative disease dynamics as a tool for guiding response to avian influenza in poultry in the United States of America. Prev Vet Med. 2014; 113(4):376-397. https://dx.doi.org/10.1016%2Fj.prevetmed.2013.11.011
- Kleiboeker SB. Swine fever: classical swine fever and African swine fever. Vet Clin North Am Food Anim Pract. 2002; 18(3):431-451. https://doi.org/10.1016/s0749-0720(02)00028-2
- Blome S, Staubach C, Henke J, Carlson J, Beer M. Classical Swine Fever-An Updated Review. Viruses. 2017; 9(4):86. https://doi.org/10.3390/v9040086
- Vargas Teran M, Calcagno Ferrat N, Lubroth J. Situation of classical swine fever and the epidemiologic and ecologic aspects affecting its distribution in the American continent. Ann N Y Acad Sci. 2004; 1026:54-64. https://doi.org/10.1196/annals.1307.007
- OIE. OIE Official disease status - Classical Swine Fever. World Organisation for Animal Health; 2020. https://www.oie.int/en/animal-health-in-the-world/official-disease-status/
- Munoz-Gonzalez S, Perez-Simo M, Munoz M, Bohorquez JA, Rosell R, Summerfield A, et al. Efficacy of a live attenuated vaccine in classical swine fever virus postnatally persistently infected pigs. Vet Res. 2015; 46:78. https://doi.org/10.1186/s13567-015-0209-9
- Martinez-Gutierrez M, Ruiz-Saenz J. Diversity of susceptible hosts in canine distemper virus infection: a systematic review and data synthesis. BMC Vet Res. 2016; 12:78. https://doi.org/10.1186/s12917-016-0702-z
- Espinal MA, Diaz FJ, Ruiz-Saenz J. Phylogenetic evidence of a new canine distemper virus lineage among domestic dogs in Colombia, South America. Vet Microbiol. 2014; 172(1-2):168-176. https://doi.org/10.1016/j.vetmic.2014.05.019
- Duque-Valencia J, Diaz FJ, Ruiz-Saenz J. Phylogenomic Analysis of Two Co-Circulating Canine Distemper Virus Lineages in Colombia. Pathogens. 2020; 9(1):26. https://doi.org/10.3390/pathogens9010026
- Duque-Garcia Y, Echeverri-Zuluaga M, Trejos-Suarez J, Ruiz-Saenz J. Prevalence and molecular epidemiology of Canine parvovirus 2 in diarrheic dogs in Colombia, South America: A possible new CPV-2a is emerging? Vet Microbiol. 2017;201:56-61. https://doi.org/10.1016/j.vetmic.2016.12.039
- Zhou P, Zeng W, Zhang X, Li S. The genetic evolution of canine parvovirus - A new perspective. PLoS One. 2017; 12(3):e0175035. https://doi.org/10.1371/journal.pone.0175035
- Rendon-Marin S, da Fontoura Budaszewski R, Canal CW, Ruiz-Saenz J. Tropism and molecular pathogenesis of canine distemper virus. Virol J. 2019; 16(1):30. https://doi.org/10.1186/s12985-019-1136-6
- Otsuki N, Nakatsu Y, Kubota T, Sekizuka T, Seki F, Sakai K, et al. The V protein of canine distemper virus is required for virus replication in human epithelial cells. PloS one. 2013; 8(12):e82343. https://doi.org/10.1371/journal.pone.0082343
- Quintero-Gil C, Rendon-Marin S, Martinez-Gutierrez M, Ruiz-Saenz J. Origin of Canine Distemper Virus: Consolidating Evidence to Understand Potential Zoonoses. Front Microbiol. 2019; 10:1982. https://doi.org/10.3389/fmicb.2019.01982
- Day MJ, Horzinek MC, Schultz RD, Squires RA, Vaccination Guidelines Group of the World Small Animal Veterinary A. WSAVA Guidelines for the vaccination of dogs and cats. J Small Anim Pract. 2016; 57(1):E1-E45. https://doi.org/10.1111/jsap.2_12431
- WHO. WHO Expert Consultation on Rabies: second report: World Health Organization; Geneva, Switzerland. 2013. https://apps.who.int/iris/handle/10665/85346
- WHO. WHO Expert Consultation on Rabies: Third report: World Health Organization; Geneva, Switzerland. 2018. https://apps.who.int/iris/handle/10665/272364
- Páez A, Hernández C, Escobar H, Zapata JJ, Méndez J, Rey-Benito G. Evaluación de la seroconversión como respuesta a la vacunación antirrabica en perros en el departamento del Valle del Cauca, Colombia, 2009. Biomédica: Revista del Instituto Nacional de Salud. 2011; 31(4):474-484. https://doi.org/10.7705/biomedica.v31i4.395
- Del Rio Vilas VJ, Freire de Carvalho MJ, Vigilato MA, Rocha F, Vokaty A, Pompei JA, et al. Tribulations of the Last Mile: Sides from a Regional Program. Front Vet Sci. 2017; 4:4. https://doi.org/10.3389/fvets.2017.00004
- Freire de Carvalho M, Vigilato MAN, Pompei JA, Rocha F, Vokaty A, Molina-Flores B, et al. Rabies in the Americas: 1998-2014. PLoS Negl Trop Dis. 2018; 12(3):e0006271. https://doi.org/10.1371/journal.pntd.0006271
- Escobar LE, Peterson AT, Favi M, Yung V, Medina-Vogel G. Bat-borne rabies in Latin America. Rev Inst Med Trop Sao Paulo. 2015; 57(1):63-72. https://doi.org/10.1590/S0036-46652015000100009
- Streicker DG, Winternitz JC, Satterfield DA, Condori-Condori RE, Broos A, Tello C, et al. Host-pathogen evolutionary signatures reveal dynamics and future invasions of vampire bat rabies. Proc Natl Acad Sci U S A. 2016; 113(39):10926-109231. https://doi.org/10.1073/pnas.1606587113
- Johnson N, Montano Hirose JA. The impact of paralytic bovine rabies transmitted by vampire bats in Latin America and the Caribbean. Rev Sci Tech. 2018; 37(2):451-459. https://doi.org/10.20506/rst.37.2.2814
- Birhane MG, Cleaton JM, Monroe BP, Wadhwa A, Orciari LA, Yager P, et al. Rabies surveillance in the United States during 2015. J Am Vet Med Assoc. 2017; 250(10):1117-1130. https://doi.org/10.2460/javma.250.10.1117
- Favoretto SR, de Mattos CC, de Mattos CA, Campos AC, Sacramento DR, Durigon EL. The emergence of wildlife species as a source of human rabies infection in Brazil. Epidemiol Infect. 2013; 141(7):1552-1561. https://doi.org/10.1017/S0950268813000198
- Arechiga-Ceballos N, Aguilar-Setien A. Alphaviral equine encephalomyelitis (Eastern, Western and Venezuelan). Rev Sci Tech. 2015; 34(2):491-501. https://doi.org/10.20506/rst.34.2.2374
- Dupuy LC, Richards MJ, Livingston BD, Hannaman D, Schmaljohn CS. A Multiagent Alphavirus DNA Vaccine Delivered by Intramuscular Electroporation Elicits Robust and Durable Virus-Specific Immune Responses in Mice and Rabbits and Completely Protects Mice against Lethal Venezuelan, Western, and Eastern Equine Encephalitis Virus Aerosol Challenges. J Immunol Res. 2018; 2018:8521060. https://doi.org/10.1155/2018/8521060
- Aguilar PV, Adams AP, Suarez V, Beingolea L, Vargas J, Manock S, et al. Genetic characterization of Venezuelan equine encephalitis virus from Bolivia, Ecuador and Peru: identification of a new subtype ID lineage. PLoS Negl Trop Dis. 2009; 3(9):e514. https://doi.org/10.1371/journal.pntd.0000514
- Ferro C, Boshell J, Moncayo AC, Gonzalez M, Ahumada ML, Kang W, et al. Natural enzootic vectors of Venezuelan equine encephalitis virus, Magdalena Valley, Colombia. Emerg Infect Dis. 2003; 9(1):49-54. https://doi.org/10.3201/eid0901.020136
- Weaver SC, Ferro C, Barrera R, Boshell J, Navarro JC. Venezuelan equine encephalitis. Annu Rev Entomol. 2004; 49:141-174. https://doi.org/10.1146/annurev.ento.49.061802.123422
- Navarro JC, Medina G, Vasquez C, Coffey LL, Wang E, Suarez A, et al. Postepizootic persistence of Venezuelan equine encephalitis virus, Venezuela. Emerg Infect Dis. 2005; 11(12):1907-1915. https://doi.org/10.3201/eid1112.050533
- Ferro C, De las Salas J, González M, Díaz A, Cabrera C, Flórez Z, et al. Do some conditions contribute to the reemergence of the Venezuelan equine encephalitis virus in the Colombian Alta Guajira? Biomédica. 2015; 35(1):62-72. http://doi.org/10.7705/biomedica.v35i1.2212
- Quiroz E, Aguilar PV, Cisneros J, Tesh RB, Weaver SC. Venezuelan equine encephalitis in Panama: fatal endemic disease and genetic diversity of etiologic viral strains. PLoS Negl Trop Dis. 2009; 3(6):e472. https://doi.org/10.1371/journal.pntd.0000472























